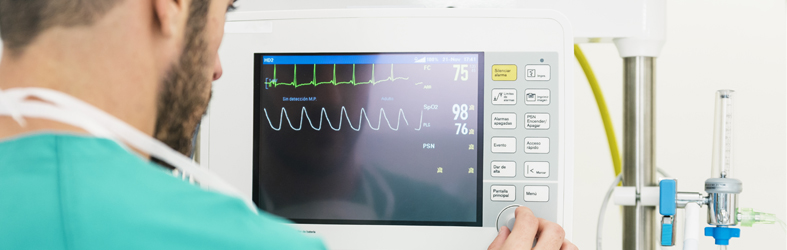
After years of development, STC has established a pre-clinical medical device R&D test platform that meets international standards, and set up a series of full-featured laboratories such as passive medical device laboratories and active medical device laboratories. We can meet the pre-clinical R&D testing needs of medical device manufacturers as well as providing one-stop service, including chemical characterization, biocompatibility testing, large animal testing, microbiological testing, safety and performance verification, electromagnetic compatibility (EMC) testing and more.
Biological Testing (ISO 10993/ GB/T 16886)
- ISO 17025 (CMA/ CNAS)
- 21 CFR Part 58 (FDA)
- OECD GLP (EMA)
Electrical Testing (IEC 60601/ GB 9706)
- ISO 17025 (CMA/ CNAS)
- CB Scheme
- NRTL Program
For more information about our Medical Devices Testing services, please feel free to contact us.
Medical Devices Testing